Five best practices for integrating drug substance and drug product development
Author:
Thermo Fisher Scientific Content Team
Category | Small molecule
Integrating drug substance and drug product development can help reduce handoffs, improve projects timelines, and support more efficient execution across development stages.
Key takeaways
- Drug substance and drug product development are interdependent and impact overall program timelines.
- Siloed execution may create coordination challenges and increase operational complexity.
- Integration can help reduce delays associated with handoffs, knowledge, and tech transfers.
- Risk identification across stages supports proactive mitigation.
- End-to-end CDMO partnerships may simplify coordination across development phases.
Drug substance and drug product development are both critical components of pharmaceutical development and manufacturing, each playing a vital role in bringing new medications to market. Drug substance development involves the synthesis or extraction of active pharmaceutical ingredients, or APIs, that are responsible for the drug's pharmacological activity. It encompasses activities such as molecular synthesis, purification, and characterization, aiming to produce a safe, pure, and effective compound that meets rigorous quality and purity standards.
- An example of a drug substance is ibuprofen, the active pharmaceutical ingredient in Advil, a nonsteroidal anti-inflammatory drug (NSAID) that reduces inflammation and pain in the body.
Conversely, drug product development involves formulating the drug substance or API into a final dosage form, such as capsules, tablets, or injections, that’s suitable for distribution and consumption. This involves carefully selecting excipients, fillers, binders, and coatings, optimizing formulations, and developing manufacturing processes to ensure the stability, bioavailability, and efficacy of the medication. Together, drug substance and drug product development are essential for translating promising drug candidates into safe and effective medications.
- An example of a drug product is an Advil-coated caplet, which contains 200 mg of ibuprofen in addition to other inactive ingredients or ancillary substances including acetylated monoglycerides and croscarmellose sodium.
Why are drug substance and drug product development siloed?
Traditionally, drug substance and drug product development have been treated as separate processes, often outsourced to different third-party vendors. This approach, while common, can lead to inefficiencies and delays in overall drug development. Separating drug substance and drug product development may result in misalignments between the two processes, such as raw material supply issues or incompatible manufacturing methods, which can impede progress and increase costs. Moreover, managing multiple vendors adds complexity to project management, communication, and coordination, potentially leading to delays.
As a result, there is a growing recognition of the need to integrate drug substance and drug product development efforts, either through in-house collaborations or through a strategic partnership with a contract development and manufacturing organization (CDMO). Integrating these activities can streamline workflows, optimize resource allocation, and accelerate the journey from molecule to medicine. In this blog, we’ll explore five best practices when integrating drug substance and drug product development and discuss why partnering with an end-to-end CDMO can prove beneficial for sponsors looking to bring new drugs to market.
Why integrate drug substance and drug product development?
By integrating drug substance and drug product development, biotechnology and pharmaceutical companies can effectively optimize processes, eliminate redundancies, and minimize delays associated with handoffs and tech transfers between different phases of drug development. Integration also facilitates early identification and mitigation of potential issues, such as formulation challenges or manufacturing constraints, leading to more informed decision-making.
Additionally, integrating drug substance and drug product development enables companies to capitalize on synergies between the two processes, allowing for greater flexibility and agility in responding to evolving project requirements. Ultimately, integration enhances collaboration, innovation, and productivity, positioning companies for success in an increasingly competitive pharmaceutical landscape while accelerating the delivery of safe and effective medications to patients.
Five best practices for integrating drug substance and drug product development
When seeking to integrate drug substance and drug product development, incorporating the following best practices into your approach is essential for optimizing efficiency and effectiveness:
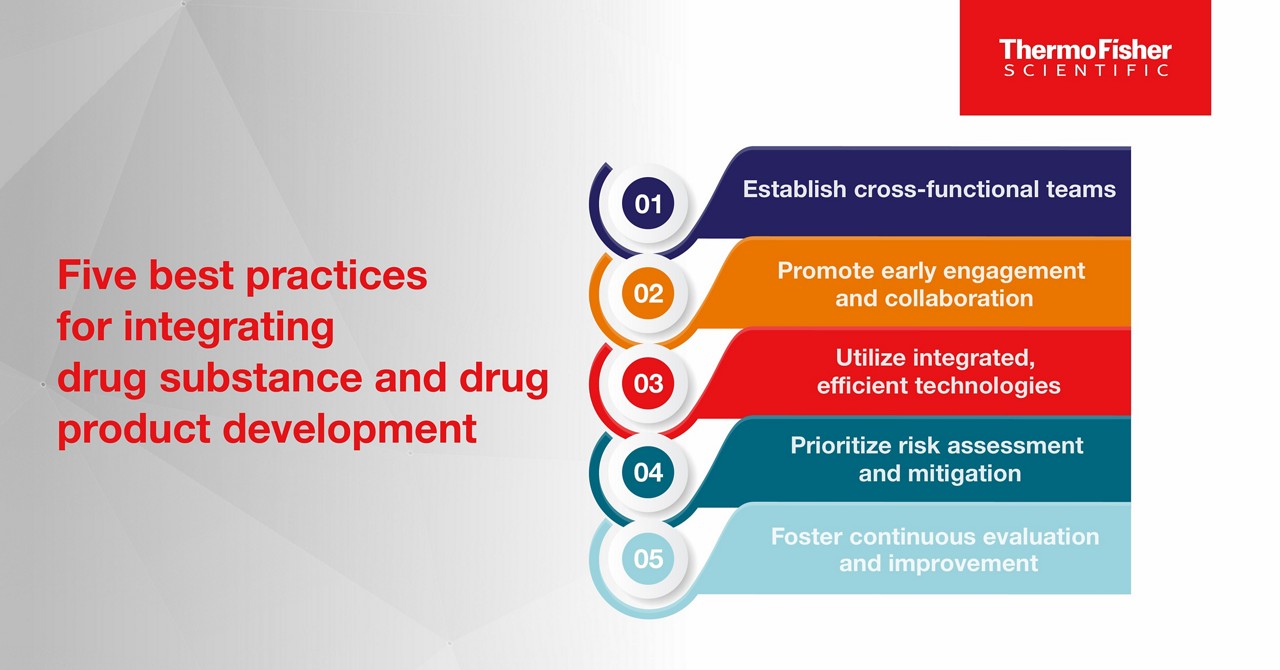
1. Establish cross-functional teams
Involving multidisciplinary experts from chemistry, formulation, regulatory affairs, manufacturing, and quality assurance teams enables a holistic approach to drug development. Cross-functional teams facilitate knowledge sharing and problem-solving, fostering an earlier understanding of challenges like stability or solubility issues. By leveraging diverse perspectives, sponsors can identify issues early on and develop contingency plans, ultimately driving efficiency.
2. Promote early engagement and collaboration
Coordinating efforts across drug substance and drug product development from the get-go allows teams to anticipate potential issues, such as formulation challenges or manufacturing constraints, and develop mitigation strategies accordingly. As soon as drug substance development teams start to synthesize APIs, their drug product development counterparts should engage in proactive problem-solving, facilitating smoother transitions between phases.
3. Utilize integrated, efficient technologies
Integrated technologies, such as cloud-based project management systems, enable real-time communication, data sharing, and task documentation, allowing teams to collaborate seamlessly. By centralizing information and resources, these technologies support decision-making and enhance transparency and traceability throughout the drug development process. Additionally, integrated technologies automate routine tasks and reduce manual errors, increasing productivity.
4. Prioritize risk assessment and mitigation
By conducting risk assessments throughout drug substance development, teams can identify challenges that may arise later in drug product development. These risks could include formulation issues, supply chain concerns, manufacturing complexities, regulatory hurdles, or safety considerations. Additionally, this approach enables teams to focus on addressing the most critical risks that have the highest potential impact on patient safety and product quality.
5. Foster continuous evaluation and improvement
Through ongoing evaluation and improvement, sponsors can adapt to changing requirements and address emerging challenges. Regularly reviewing project progress, soliciting feedback from teams, evaluating performance metrics, and identifying areas for improvement enable sponsors to streamline workflows and improve efficiency. What’s more, it allows teams to capitalize on lessons learned from previous experiences and optimize methods moving forward.
How can CDMOs help integrate drug substance and drug product development?
CDMOs can play a crucial role in integrating drug substance and drug product development by providing comprehensive services that cover the entire spectrum of pharmaceutical manufacturing. These integrated, end-to-end companies bring together expertise in chemistry, formulation, analytical testing, and manufacturing, offering biotech and pharma partners access to specialized capabilities and resources not typically available in-house.
Consolidating services such as API synthesis, formulation development, process optimization, scale-up, and regulatory support under one roof through a single CDMO partner simplifies the complexities and challenges associated with coordinating with multiple vendors. By serving as a strategic partner, these CDMOs can facilitate the seamless integration of drug substance and drug product development activities from concept to commercialization.
Summary
Integrating drug substance and drug product development may help reduce fragmentation, improve timelines and coordination, and support more efficient execution across the development lifecycle. By establishing cross-functional teams, engaging early across functions, leveraging integrated technologies, prioritizing risk mitigation, and fostering continuous improvement, sponsors can take a structured approach to integration while maintaining quality and regulatory alignment.
To learn how leveraging an integrated approach can help expedite the early-phase development of small molecules, read our white paper.


