Sterile fill–finish services
Efficient sterile drug manufacturing and development for injectable medicines
Sterile fill–finish is the final stage of pharmaceutical manufacturing, where drug products are aseptically filled under controlled sterile conditions into their final containers such as vials, prefilled syringes, or cartridges. Thermo Fisher Scientific provides sterile fill–finish services that support biologics and small molecules from development through commercial manufacturing, helping sponsors reduce risk, meet GMP requirements, and deliver safe parenteral medicines to patients.
Thermo Fisher’s CGMP facilities accommodate biologics, small molecules, and mRNA therapeutics with flexible sterile fill-finish services from first-in-human through commercial scale. Non-CGMP options support preclinical work with scalable equipment configurations. Global regulatory teams guide projects to successful NDA and BLA approval across US and EU markets, with manufacturing sites strategically located across North America, Europe, and Asia-Pacific.
Full lifecycle support from first-in-human to commercial scale
130+ million sterile liquid and lyophilized vials produced annually
Global manufacturing network across US, EU, and Singapore
cGMP and non-cGMP options for various development stages
What sterile dosage formats do you support?
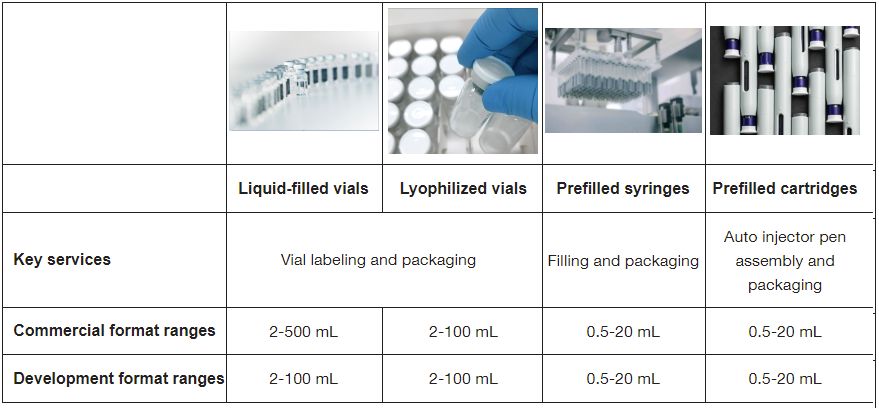
Our sterile fill–finish manufacturing services support four primary container formats. Each format accommodates specific molecule requirements and scales seamlessly from preclinical through commercial production.
- Liquid-filled vials: 2–500 mL commercial and 2–100 mL clinical format ranges
- Lyophilized vials: 2–100 mL commercial and clinical format ranges
- Prefilled syringes: 0.5–20 mL commercial and clinical format ranges
- Prefilled cartridges: 0.5–20 mL commercial and clinical format ranges
The table below details specifications for each parenteral dosage forms across development and commercial scales.
 |
 |
 |
 |
|
| Key services | ||||
| Commercial format ranges | ||||
| Development format ranges | ||||
Ready to discuss your sterile fill-finish project? Contact our team or Download our steriles brochure to explore our US and EU development capabilitites.
Aseptic fill-finish solutions for parenteral drug products
Thermo Fisher provides integrated aseptic fill–finish solutions that support parenteral drug products from formulation development through clinical and commercial manufacturing. Our teams work across formulation, sterile manufacturing, packaging, analytical development, and cold chain distribution—helping ensure products are developed, filled, finished, and delivered with the quality, consistency, and regulatory readiness required at each stage of the lifecycle.
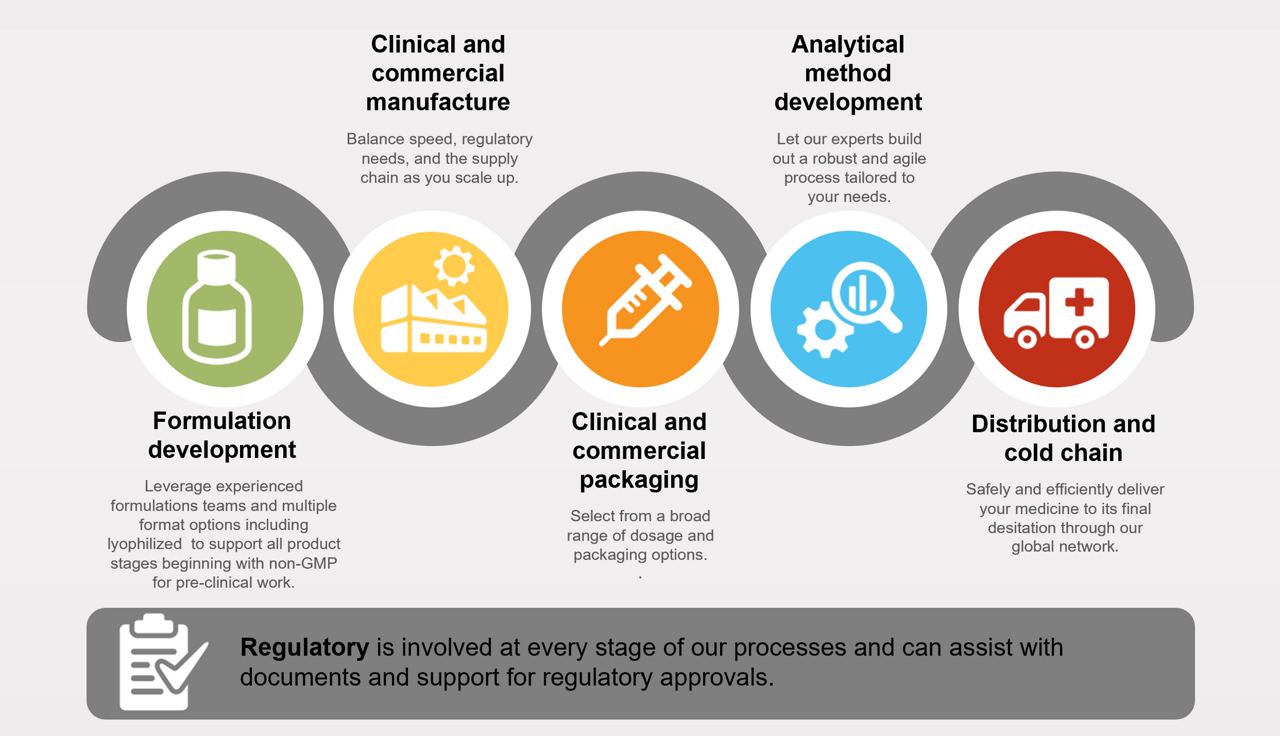
Developing the correct formulation can be time-consuming when delays are least affordable. The chemical nature of parenteral compounds may present challenges such as temperature and shear sensitivity, poor solubility, and high potency. Lyophilization, when required, demands careful cycle optimization to support downstream sterile fill–finish and scale-up.
These formulation activities are designed to prepare parenteral drug products for reliable sterile manufacturing.
Our established parenteral formulation processes include:
- Pre‑formulation: Evaluation of optimal pH, ionic strength, degradation pathways, and stability
- Formulation development: Selection and definition of optimal excipient levels using OFaT and/or design of experiments (DoE)
- Formulation confirmation: Stability confirmation of the final formulation using analytical tools
Formulation support is available for:

Reducing cost and risk in preclinical and Phase I studies
Preclinical and Phase I studies can have lengthy timelines that lead to increased costs. Ensuring quality and consistency across processes in early development is critical as you move from phase to phase.
Lyophilization, use of non-CGMP batches, and pre-qualified components can address both concerns. Lyophilization increases stability, leading to a longer shelf-life for the duration of clinical trials.
To save time and money, we offer non-CGMP suites that ensure scalable equipment. The setup of our non-CGMP lines mimics our CGMP lines—it’s the same material without the aseptic processing and costs associated with CGMP spaces.
Successfully scaling sterile drug products requires balancing speed, regulatory requirements, and supply chain reliability. We support clinical‑to‑commercial sterile fill–finish manufacturing within a global network.
We help you:
- Navigate complex aseptic manufacturing regulatory environments at a global scale
- Accelerate product availability for patients
- Execute sterile fill–finish technology transfers efficiently
- Build a robust, resilient global supply chain
Thermo Fisher’s experienced regulatory teams support drug commercialization through integrated clinical trial solutions, global technical expertise, regulatory insight, and coordinated transportation.

Whether you have a defined dosage format or are evaluating options, Thermo Fisher offers a wide range of packaging solutions. In a single year, we produced more than 130 million sterile liquid and lyophilized vials and manufactured 40 dosage forms, supported by pre‑qualified components and standardized processes.
Sterile drug product packaging options include:
- Small‑ and large‑volume parenterals
- Liquid‑filled vials
- Lyophilized vials
- Extensive vial sizes, including ISO standards
- Prefilled syringes and cartridges
Pre‑qualified components and standardized processes
Thermo Fisher’s selection of product contact materials and components can shorten timelines, reduce capital expenditures, and eliminate cleaning validation, media fills, and container closure integrity (CCI).
Our analytical and stability services are integrated with sterile fill–finish process development and scale‑up, supporting product performance across the lifecycle.
Capabilities include:
- Lyo-cycle process development and optimization
- Manufacturing process development studies:
- Mixing and pump-shearing
- Hold time studies
- Freeze-thaw studies
- Scalability studies
- Product contact part compatibility
- Proven Acceptable Ranges (PARs)
- International Council for Humanization (ICH) stability
- Shipping container selection
- Component compatibility testing
- Cleaning validation
- In-use stability
- Sterilization cycle development and validation

With more than 30 years of clinical trial experience, Thermo Fisher supports safe, on‑time delivery of sterile and injectable therapies worldwide. Services span strategy, sourcing, management, packaging and labeling, storage, and global distribution for small molecules, biologics, and advanced therapies.
For temperature‑sensitive sterile products, cold chain integrity is critical.
Our cold chain services provide dependable site‑to‑site connectivity through a global network offering storage, packaging, labeling, and shipment capabilities designed to maintain product integrity through delivery.
Learn more about our cold and ultra-cold supply chain management and logistics services.

Why choose Thermo Fisher for sterile fill–finish?
- Global sterile manufacturing network supporting clinical and commercial supply
- Experience across sterile dosage forms, including vials, syringes, and cartridges
- Integrated CDMO model from development through commercialization
- Regulatory and GMP expertise supporting global submissions and inspections
Frequently asked questions about sterile fill–finish services
Sterile fill–finish manufacturing is the aseptic process of filling drug products into final containers, such as vials or syringes, under controlled conditions to prevent contamination and ensure patient safety.
Our facilities operate under GMP requirements for sterile pharmaceutical products, using validated aseptic processes, qualified equipment, and robust quality systems.
Yes. Sterile fill–finish services support biologics, small molecules, and other complex injectable drug products requiring aseptic processing.
Lyophilization, or freeze-drying, is a sterile fill–finish process that removes water from drug products to enhance stability and extend shelf life. This technique benefits heat-sensitive biologics and small molecules by preserving molecular integrity during storage and transportation, reducing the need for cold chain requirements.
Our sterile fill-finish global site locations
Our 642,000 sq. ft. campus in Monza, Italy is a center of excellence for sterile manufacturing. It features a unique co-location of mRNA manufacturing capabilities with LNP and fill-finish services to help streamline your processes and mitigate risks.
Our Greenville, NC facility is a large, multipurpose pharmaceutical manufacturing and packaging campus. This site provides both solid dose form manufacturing and packaging and sterile dose manufacturing, filling, and lyophilization of both biopharmaceuticals and small molecules.
Our Ferentino facility is 14,034 sq. m. (151,061 sq. ft.), specializing in integrated sterile liquid and lyophilized product development and commercial manufacturing, including high-potency products and LVP.
Our Swindon facility specializes in integrated sterile liquid and lyophilized product development and commercial manufacturing, including high-potency products and LVP. It offers extensive development and commercial capabilities for sterile dosage forms, including liquid vials, commercial fill-finish lines, and primary and secondary packaging vials.
Our state-of-the-art sterile drug development and manufacturing facility in Singapore supports rapid fill-finish vaccines and other therapeutics for an expanding biopharma market.
Helpful resources


